LL-37 Peptide: The Body’s Built-In Defense and Regeneration Signal

Among the many fascinating molecules produced naturally by the human body, few are as multifunctional and powerful as LL-37. Classified as an antimicrobial peptide, LL-37 plays a central role in immune defense, inflammation regulation, and tissue repair. As research continues to expand, this peptide has drawn attention in immunology, dermatology, regenerative medicine, and even oncology.
What Is LL-37?
LL-37 is the only known human member of the cathelicidin family of antimicrobial peptides. It is derived from a precursor protein called hCAP-18 (human cationic antimicrobial protein-18), which is cleaved to form the active peptide LL-37. The name “LL-37” refers to its structure: it begins with two leucine residues (“LL”) and consists of 37 amino acids.
This peptide is produced by various cells in the body, including neutrophils, macrophages, epithelial cells, and keratinocytes. It is found in tissues and fluids such as skin, lungs, gastrointestinal tract, saliva, and sweat—strategic locations where pathogens commonly attempt to enter the body.
Antimicrobial Power
LL-37’s primary role is as part of the innate immune system—the body’s first line of defense. Unlike antibodies, which are highly specific and require prior exposure, LL-37 acts rapidly and broadly.
It works by disrupting microbial membranes. Because many bacteria, viruses, and fungi have negatively charged membranes, LL-37—being positively charged—binds to them and destabilizes their structure. This can lead to direct microbial death.
Research shows LL-37 has activity against:
- Gram-positive bacteria
-
Gram-negative bacteria
-
Certain viruses
-
Fungal pathogens
Importantly, LL-37’s mechanism makes it difficult for microbes to develop resistance, which has fueled interest in it as a potential therapeutic template.
Immune Modulation
Beyond direct antimicrobial effects, LL-37 acts as an immune signaling molecule. It helps regulate inflammation rather than simply triggering it.
LL-37 can:
-
Attract immune cells to sites of infection (chemotaxis)
-
Stimulate cytokine production
-
Enhance wound healing responses
-
Modulate inflammatory signaling pathways
This dual role—killing pathogens while shaping immune response—makes LL-37 particularly unique. It does not simply attack invaders; it coordinates the body’s broader defense strategy.
However, balance is critical. Excessive LL-37 expression has been linked to inflammatory conditions such as psoriasis, where it may contribute to immune system overactivation.
Role in Wound Healing and Tissue Repair
LL-37 also plays a significant role in tissue regeneration. It promotes:
-
Angiogenesis (formation of new blood vessels)
-
Keratinocyte migration
-
Re-epithelialization of wounds
-
Collagen production
These properties have led researchers to explore LL-37 in the context of chronic wounds, diabetic ulcers, and skin injuries.
Because it supports both antimicrobial defense and tissue repair, LL-37 represents a natural bridge between immune protection and regenerative biology.
LL-37 and Vitamin D
One of the most interesting aspects of LL-37 biology is its connection to vitamin D. Activation of the vitamin D receptor stimulates production of cathelicidin, including LL-37.
This relationship has sparked investigations into how vitamin D levels may influence immune resilience. Some researchers propose that adequate vitamin D status supports innate immunity partly by enhancing LL-37 expression.
This link has been studied in respiratory infections and immune health, though clinical applications are still under active investigation.
Potential Therapeutic Applications
Because LL-37 has antimicrobial, anti-biofilm, regenerative, and immune-modulating properties, scientists are studying it in multiple areas:
1. Chronic infections:
Its ability to disrupt biofilms makes it a candidate for hard-to-treat bacterial infections.
2. Dermatology:
Topical applications are being explored for wound healing and skin conditions.
3. Cancer research:
Some studies suggest LL-37 may influence tumor growth—either promoting or inhibiting it depending on context. This dual behavior means much more research is needed.
4. Sepsis and immune dysregulation:
Its immune-modulating properties make it a molecule of interest in inflammatory disorders.
It’s important to note that while laboratory and early-stage research is promising, LL-37 is still largely in experimental phases for many applications.
Risks and Considerations
As with many biologically active molecules, context determines outcome. LL-37 can be protective in one setting and harmful in another. Elevated levels have been associated with autoimmune and inflammatory conditions such as:
-
Psoriasis
-
Rosacea
-
Certain lupus-related processes
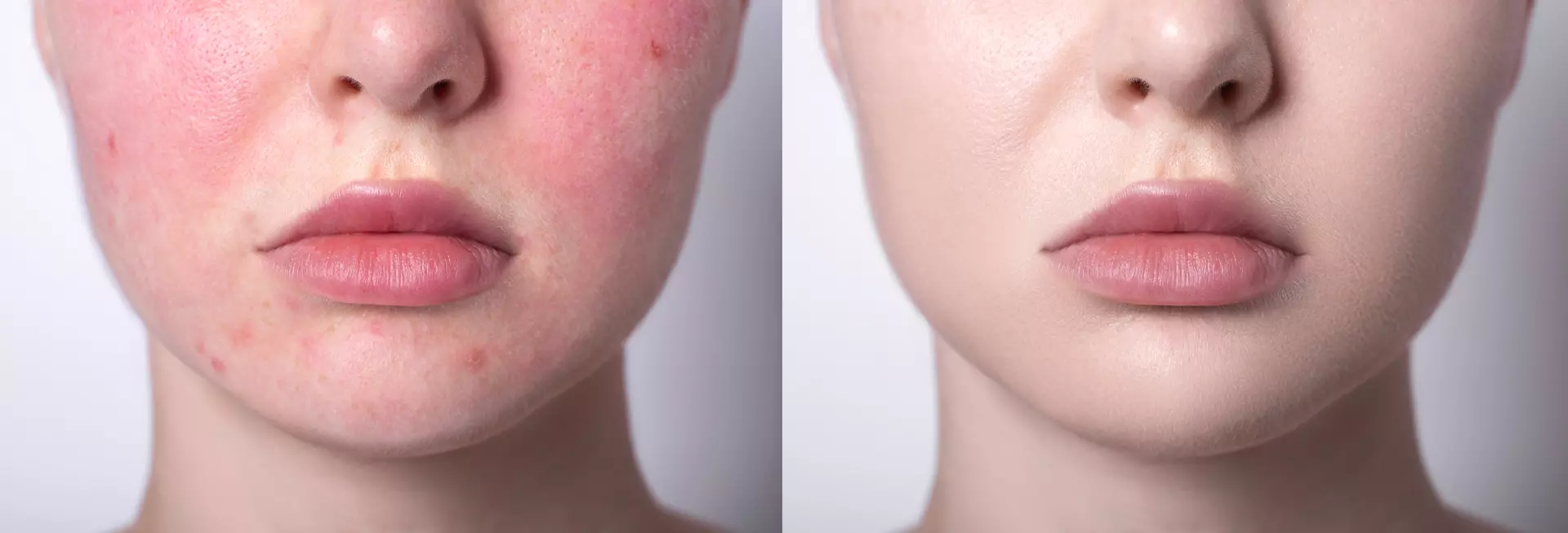
This underscores the complexity of immune peptides. They are not “good” or “bad” in isolation—they are regulatory signals that must operate within precise biological balance.
The Bigger Picture
LL-37 represents a broader category of molecules known as host defense peptides. These peptides are evolutionarily conserved and form a critical part of innate immunity across species.
What makes LL-37 especially compelling is its versatility:
-
Direct antimicrobial action
-
Immune signaling
-
Tissue regeneration
-
Interaction with vitamin D pathways
-
Involvement in inflammatory disease
Rather than functioning as a simple antibiotic, LL-37 acts as a biological conductor, coordinating multiple layers of defense and repair.
Final Thoughts
LL-37 is a powerful example of the sophistication of the human immune system. It operates at the intersection of microbiology, immunology, and regenerative medicine. While still under active study, it offers promising insights into how the body naturally protects and heals itself.
As research advances, LL-37 may inspire new therapeutic approaches that harness the body’s own defense strategies rather than relying solely on synthetic drugs. For now, it remains one of the most fascinating peptides in human biology—a small molecule with remarkably broad influence.
























